Systematic Chemical Naming Guide | Accurate IUPAC Names Explained 2026
Systematic chemical naming is something students, researchers, and professionals rely on when they want to identify a compound with complete scientific accuracy.
In my thoughts, knowing these core points helps you master systematic naming confidently and apply it effectively in real chemical work.
The following sections expand on these key points and guide you toward the choices.
Basics of IUPAC Naming 🧪

Chemists use IUPAC rules to create a standard name for every molecule.
These rules keep naming clear in every country.
This section covers core ideas you must know first.
• Longest Chain Rule
Meaning: Choose the longest carbon chain as the parent name.
• Parent Hydrocarbon
Meaning: Base name like methane, ethane, propane.
• Numbering Direction
Meaning: Number from the end closest to a substituent.
• Locant Rule
Meaning: Use numbers to show where each group sits.
• Lowest Set Rule
Meaning: Give substituents the lowest numbers possible.
• Alphabetical Order
Meaning: Name substituents alphabetically, ignoring prefixes.
• Multiplying Prefixes
Meaning: Use di-, tri-, tetra- for repeating groups.
• Simple Suffixes
Meaning: Use -ane, -ene, -yne for single, double, triple bonds.
• Functional Group Priority
Meaning: Some groups outrank others for suffix naming.
• Final Assembly Rule
Meaning: Combine prefix + parent + suffix in order.
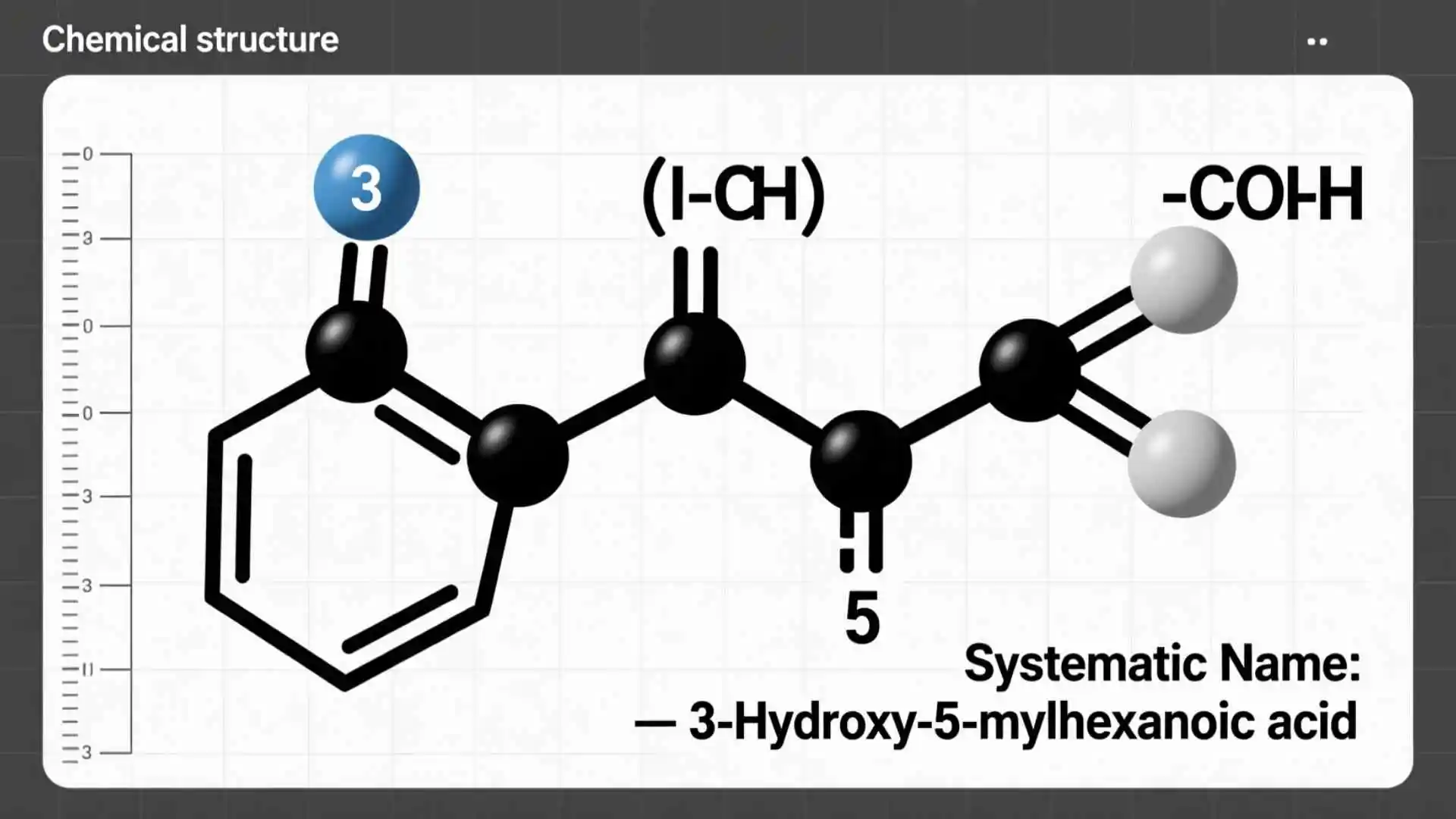
Provide the correct systematic name for the compound shown here
This query often appears in chemistry homework and exams when students must identify the official IUPAC name of a compound from its formula or structure.
Understanding naming rules helps learners quickly recognize how atoms combine and how prefixes describe the number of atoms.
• Dinitrogen Trioxide
Meaning: The correct IUPAC systematic name for N₂O₃.
• Nitrogen(III) Oxide
Meaning: Indicates nitrogen has an oxidation state of +3.
• Binary Molecular Oxide
Meaning: A compound made of two non-metals, nitrogen and oxygen.
• Two-Nitrogen Three-Oxygen Compound
Meaning: Describes the exact atom count in the formula.
• Prefix-Based Nitrogen Oxide
Meaning: Uses the Greek prefixes “di” and “tri” for naming.
• Covalent Nitrogen Oxide
Meaning: Nitrogen and oxygen share electrons in this compound.
• Low-Temperature Oxide
Meaning: The compound stays stable mainly at cooler temperatures.
• Nitrogen Pair Oxide
Meaning: Highlights the presence of two nitrogen atoms bonded in the structure.
• Tri-Oxygen Nitrogen Compound
Meaning: Focuses on the three oxygen atoms in the molecule.
• Acid-Forming Oxide
Meaning: This oxide forms nitrous acid when it reacts with water.
Provide the correct systematic name for the compound shown here che
Students search this phrase when they want the precise chemical name that follows IUPAC rules instead of a common or informal name.
Learning systematic names improves understanding of molecular structure and chemical relationships.
• Dinitrogen Trioxide Identifier
Meaning: The recognized scientific label for the N₂O₃ molecule.
• Nitrogen Oxide with Di-Tri Prefix
Meaning: Uses Greek prefixes to indicate atom numbers.
• Covalent Nitrogen-Oxygen Molecule
Meaning: Nitrogen and oxygen atoms bond through shared electrons.
• Nitrogen Oxide Family Member
Meaning: Belongs to a group of compounds formed by nitrogen and oxygen.
• Three-Oxygen Nitrogen Compound
Meaning: A nitrogen oxide containing three oxygen atoms.
• Molecular Nitrogen Oxide
Meaning: Exists as a discrete molecule rather than an ionic compound.
• Intermediate Nitrogen Oxide
Meaning: Appears between other nitrogen oxides in oxidation level.
• Nitrous Acid Precursor
Meaning: Forms nitrous acid when dissolved in water.
• Balanced Nitrogen Oxide Structure
Meaning: Reflects the stable bonding arrangement in the molecule.
• Two-to-Three Atom Ratio Oxide
Meaning: Describes the 2:3 ratio of nitrogen to oxygen atoms.
What is the systematic name of N2O3
People often search this question when studying molecular compounds or preparing for chemistry exams.
The systematic name follows strict rules that show the number of atoms in the compound.
• Dinitrogen Trioxide
Meaning: The official IUPAC name that directly reflects the formula N₂O₃.
• Nitrogen(III) Oxide
Meaning: Shows nitrogen atoms have a +3 oxidation state.
• Di-Nitrogen Tri-Oxide Compound
Meaning: Breaks the formula into prefixes for easier learning.
• Nitrous Anhydride
Meaning: A traditional name because it forms nitrous acid with water.
• Molecular Nitrogen Oxide N₂O₃
Meaning: Emphasizes the exact molecular formula.
• Covalent Oxide of Nitrogen
Meaning: Nitrogen bonds with oxygen using shared electrons.
• Blue Liquid Nitrogen Oxide
Meaning: The compound shows a bluish color in liquid form.
• Unstable Nitrogen Oxide
Meaning: The compound easily breaks into other nitrogen oxides.
• Tri-Oxygen Nitrogen Molecule
Meaning: Focuses on the presence of three oxygen atoms.
• Nitrogen Oxide Bridge Compound
Meaning: Exists between other nitrogen oxide oxidation states.
What is the correct IUPAC name for N2O3
Many learners search this question when they want the exact IUPAC-approved chemical name used in textbooks and scientific writing.
The name helps students apply standard naming rules for molecular compounds.
• Dinitrogen Trioxide
Meaning: The precise IUPAC systematic name for N₂O₃.
• Nitrogen(III) Oxide
Meaning: Identifies nitrogen’s oxidation number as +3.
• Di-Nitrogen Molecular Oxide
Meaning: Indicates the compound contains two nitrogen atoms.
• Tri-Oxygen Molecular Oxide
Meaning: Shows that the molecule includes three oxygen atoms.
• Binary Covalent Nitrogen Oxide
Meaning: A compound formed from two non-metal elements.
• Nitrogen-Oxygen Molecular Compound
Meaning: Highlights bonding between nitrogen and oxygen atoms.
• Reactive Nitrogen Oxide
Meaning: The compound easily reacts and changes form.
• Nitrous Acid Source Oxide
Meaning: Produces nitrous acid when it reacts with water.
• Intermediate Oxidation Nitrogen Compound
Meaning: Represents a mid-level oxidation state among nitrogen oxides.
• Formula-Based Nitrogen Oxide Name
Meaning: The name directly reflects the formula N₂O₃.
Functional Groups Priority 🌡️
Functional groups have a ranking that controls naming.
This ranking chooses the suffix and numbering.
Here are important priority rules.
• Carboxylic Acid Priority
Meaning: Gets the main suffix “-oic acid.”
• Ester Priority
Meaning: Named with “-oate.”
• Aldehyde Priority
Meaning: Suffix “-al.”
• Ketone Priority
Meaning: Suffix “-one.”
• Alcohol Priority
Meaning: Suffix “-ol.”
• Amine Priority
Meaning: Suffix “-amine.”
• Amide Priority
Meaning: Suffix “-amide.”
• Nitrile Priority
Meaning: Suffix “-nitrile.”
• Alkene Ranking
Meaning: Double bonds get “-ene.”
• Alkyne Ranking
Meaning: Triple bonds get “-yne.”
Naming Alkanes 🔹

Alkanes are simple hydrocarbons.
They contain only single bonds.
These naming ideas help identify them.
• Methane Base
Meaning: One-carbon chain.
• Ethane Base
Meaning: Two-carbon chain.
• Propane Base
Meaning: Three-carbon chain.
• Butane Base
Meaning: Four-carbon chain.
• Pentane Base
Meaning: Five-carbon chain.
• Hexane Base
Meaning: Six-carbon chain.
• Heptane Base
Meaning: Seven-carbon chain.
• Octane Base
Meaning: Eight-carbon chain.
• Nonane Base
Meaning: Nine-carbon chain.
• Decane Base
Meaning: Ten-carbon chain.
Naming Substituents 🔍
Substituents attach to the main chain.
They change the name but not the parent structure.
These are common substituent details.
• Methyl Group
Meaning: –CH₃ branch.
• Ethyl Group
Meaning: –CH₂CH₃ branch.
• Propyl Group
Meaning: 3-carbon side chain.
• Isopropyl Group
Meaning: Branched 3-carbon substituent.
• Butyl Group
Meaning: 4-carbon substituent.
• Isobutyl Group
Meaning: Branched version of butyl.
• Tert-butyl Group
Meaning: T-shaped bulky group.
• Fluoro Substituent
Meaning: -F atom on chain.
• Chloro Substituent
Meaning: -Cl on chain.
• Bromo Substituent
Meaning: -Br on chain.
Naming Alkenes 🧬

Double bonds change the suffix.
Their position must be numbered correctly.
Here are rules for naming alkenes.
• Ene Suffix
Meaning: Shows presence of double bond.
• Lowest Locant
Meaning: Double bond gets lowest number.
• Substituent Before Parent
Meaning: Prefix comes before alkene name.
• Multiple Double Bonds
Meaning: Use diene, triene, etc.
• Cyclic Alkenes
Meaning: Number around ring to give lowest.
• Stereochemistry Labels
Meaning: Use E/Z system.
• Avoid Common Names
Meaning: Use systematic names only.
• Parent Chain Includes Bond
Meaning: Must include double bond.
• Handle Substitution First
Meaning: Then name the double bond.
• Longest Chain Still Wins
Meaning: But bond must be included.
Naming Alkynes ⚡
Triple bonds use “-yne.”
Their numbering follows alkene rules.
Here are alkyne naming rules.
• Yne Suffix
Meaning: Shows triple bond.
• Number Closest to Bond
Meaning: Triple bond gets priority.
• Combine with Substituents
Meaning: Prefixes apply first.
• Multiple Triple Bonds
Meaning: Use diyne, triyne.
• Mixed Unsaturation
Meaning: Use “en-yne.”
• Cyclic Alkynes
Meaning: Rare but numbered similarly.
• Avoid Hyphens Excessively
Meaning: Use proper IUPAC spacing.
• Consistent Direction
Meaning: Once you start numbering, continue.
• Priority Over Alkyl Groups
Meaning: Bond beats substituent location.
• Keep Parent Name Clear
Meaning: Never drop suffix.
Naming Aromatic Compounds 🌸

Aromatic rings have special rules.
The benzene ring often acts as the parent.
These concepts guide their naming.
• Benzene Parent Name
Meaning: Main aromatic structure.
• Substitution Numbering
Meaning: Choose lowest possible positions.
• Di-substituted Rings
Meaning: Use 1,2-; 1,3-; 1,4-.
• Avoid Ortho/Meta/Para
Meaning: Use numeric locants.
• Nitro Group
Meaning: -NO₂ substituent.
• Phenyl Group
Meaning: Benzene ring as substituent.
• Aromatic Priority Rules
Meaning: Some groups outrank others.
• Combined Substitution
Meaning: Name alphabetically.
• Fused Rings
Meaning: Special naming needed.
• Simplify Only if Allowed
Meaning: Keep IUPAC standard.
Polyfunctional Compound Naming ⚗️
Compounds with many groups need priority.
This section explains multi-group naming.
Use these rules carefully.
• Highest Priority Wins Suffix
Meaning: Others become prefixes.
• Ester Before Alcohol
Meaning: “-oate” overrides “-ol.”
• Carboxylic Acid Tops All
Meaning: Always suffix.
• Amide Over Amine
Meaning: “-amide” ranks higher.
• Aldehyde Beats Alkene
Meaning: “-al” outranks “-ene.”
• Ketone Over Alcohol
Meaning: “-one” outranks “-ol.”
• Cyano Group Ranks High
Meaning: Nitrile suffix often chosen.
• Mixed Unsaturation
Meaning: Use both “-ene” and “-yne.”
• Prefix Placement
Meaning: Put substituents first.
• Final Suffix at End
Meaning: Only one main suffix allowed.
Stereochemistry Naming 🔄

Some molecules have direction and shape.
We name them with special labels.
These rules help identify arrangement.
• R/S System
Meaning: Shows chiral center direction.
• E/Z System
Meaning: Double-bond orientation.
• Cis/Trans Only for Simple Cases
Meaning: Use sparingly.
• Wedge and Dash Rules
Meaning: 3D structure.
• Numbering Centers
Meaning: Label positions correctly.
• Mirror Images
Meaning: Enantiomers.
• Non-mirror Pairs
Meaning: Diastereomers.
• Rotation Not Important
Meaning: Use structure, not looks.
• Use Brackets
Meaning: Put (R) or (S) before name.
• Combine with Suffixes
Meaning: Works in final full name.
How to Name the Compound Correctly 🎯
Now you know all rules.
Naming the compound means applying them in order.
Here is the simple process.
• Identify Longest Chain
Meaning: Choose parent carbon line.
• Find Functional Groups
Meaning: Note all groups present.
• Rank Priority
Meaning: Choose main suffix.
• Number the Chain
Meaning: Start closest to priority group.
• Locate Substituents
Meaning: Add numbers.
• Add Prefixes
Meaning: Alphabetical order.
• Insert Unsaturation Positions
Meaning: For double or triple bonds.
• Add Stereochemistry
Meaning: R/S or E/Z.
• Combine Everything
Meaning: Prefix + parent + suffix.
• Final Check
Meaning: Ensure lowest locants.
FAQs
1. What is the systematic name of the compound Mn₃(PO₄)₂?
The systematic name of Mn₃(PO₄)₂ is manganese(II) phosphate. In this compound, phosphate (PO₄³⁻) carries a −3 charge, and two phosphate ions contribute −6 total charge. To balance this, three manganese ions must each have a +2 charge. Therefore, the correct systematic name uses the oxidation state: manganese(II) phosphate.
2. What is the systematic name of the following compound Mn₃(PO₄)₂?
The compound Mn₃(PO₄)₂ is systematically named manganese(II) phosphate. Chemists determine this by analyzing ionic charges. Phosphate ions have a −3 charge, and when two are present the total becomes −6. Three manganese atoms balance the charge with +2 each, giving the compound its proper systematic name.
3. What is the IUPAC name of the compound Mn₃(PO₄)₂?
The IUPAC name of Mn₃(PO₄)₂ is manganese(II) phosphate. IUPAC naming rules require indicating the oxidation state of transition metals using Roman numerals. Since manganese has a +2 oxidation state in this compound, the name clearly identifies it as manganese(II) phosphate combined with phosphate ions.
4. Why does Mn₃(PO₄)₂ use the Roman numeral (II) in its name?
The Roman numeral (II) indicates the oxidation state of manganese in the compound. Manganese is a transition metal that can have multiple oxidation states. In Mn₃(PO₄)₂, each manganese ion has a +2 charge, which balances the −6 charge from two phosphate ions, so the compound is named manganese(II) phosphate.
5. What is the systematic name of Li₂S?
The systematic name of Li₂S is lithium sulfide. Lithium is an alkali metal that always forms a +1 ion (Li⁺), while sulfur forms a −2 ion (S²⁻). Two lithium ions are required to balance one sulfide ion, giving the formula Li₂S and the straightforward name lithium sulfide.
6. Why doesn’t lithium sulfide need Roman numerals in its name?
Lithium sulfide does not require Roman numerals because lithium only forms one common oxidation state: +1. Unlike transition metals, it does not have multiple possible charges. Because of this fixed charge, the compound Li₂S is simply named lithium sulfide without indicating oxidation numbers.
7. How do you determine the systematic name of ionic compounds?
To determine a systematic name, first identify the cation (positive ion) and anion (negative ion). Then check their charges and balance them to understand oxidation states. For transition metals, include Roman numerals to show the metal’s charge. Finally, write the metal name first and the nonmetal or polyatomic ion second.
Conclusion
You can name any compound when you follow IUPAC rules, because every rule builds a clear path, and each step makes the name precise so you never guess again.
Discover More Topics:
- 159+ Good Name for a Black Cat | Cute, Magical & Unique Ideas 2026
- 189+ Childrens Name Tattoos for Dads | Meaningful & Stylish Ideas 2026
- 139+ Neon Name Sign for Wedding | Romantic, Modern & Aesthetic Ideas 2026

Emily Rhodes is a contemporary fiction author known for emotionally engaging stories that explore love, identity, and quiet resilience through lyrical storytelling.
Books by Emily Rhodes
-
Where the Quiet Hearts Rest
-
Beneath the Light We Hide